Nicholas Tjahjono, a fourth-year doctoral student in our group and a NASA Space Technology Graduate Research Opportunities Fellow has been awarded the Outstanding Teaching Assistant Award of the Rice’s MSNE Department.
Tjahjono’s research aims to aid in NASA’s Artemis mission to the moon and beyond. His objective is to develop mechanically robust and self-healing materials capable of operating under, and protecting astronauts from, extreme space environments such as extreme heat and cold, bombardment from micrometeoroids and orbital debris, and galactic cosmic radiation.


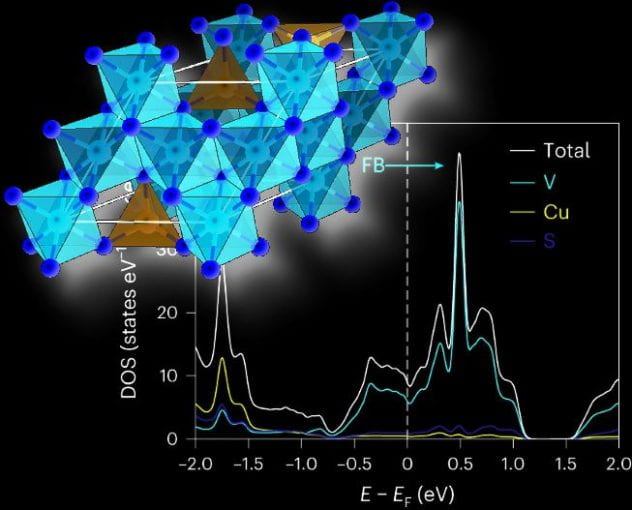


 contoured substrates
contoured substrates
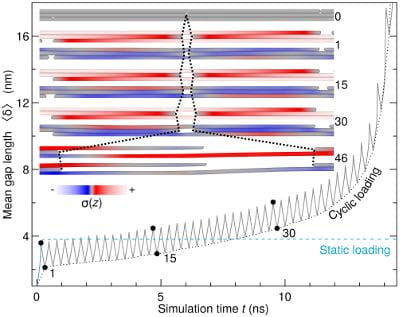 Up here in the macro world, we all feel fatigue now and then. It’s the same for bundles of
Up here in the macro world, we all feel fatigue now and then. It’s the same for bundles of 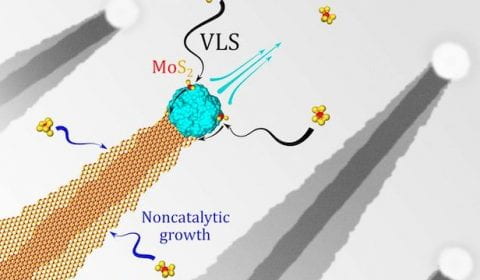 It’s now possible to quickly make ultrathin nanoribbons of
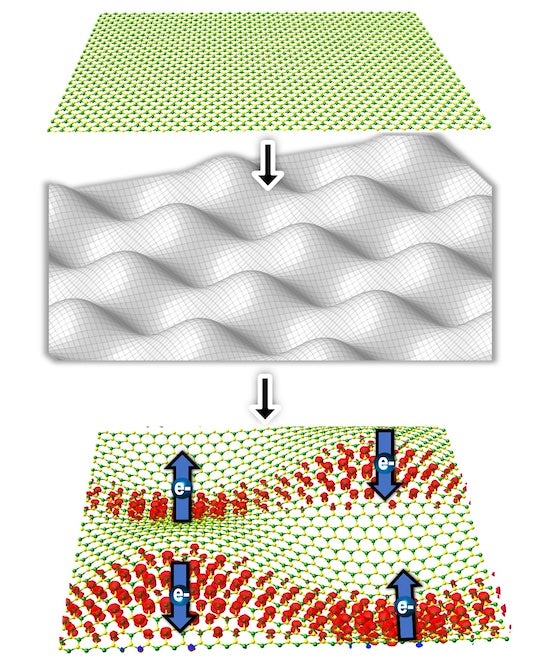
It’s now possible to quickly make ultrathin nanoribbons of 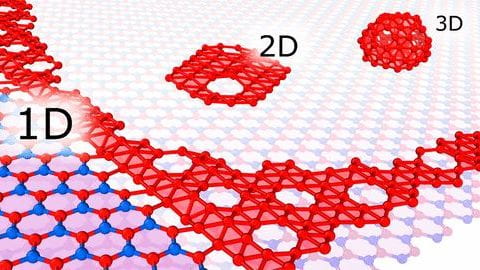
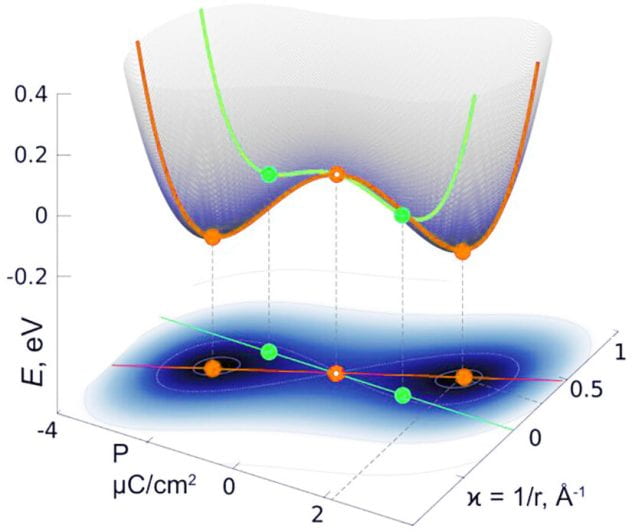
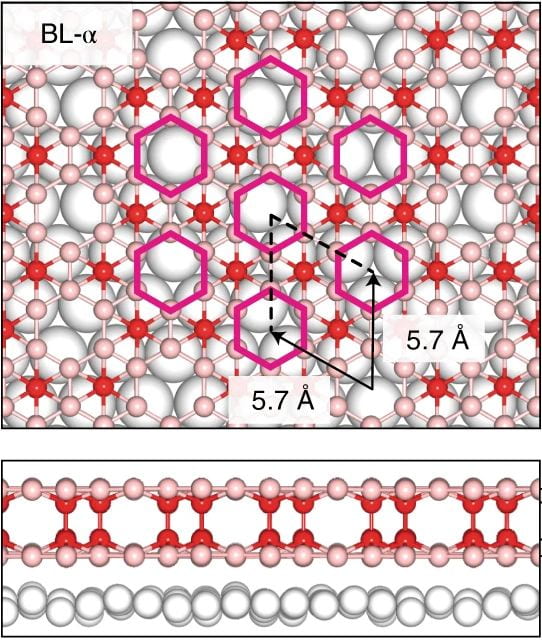 If one layer of
If one layer of 
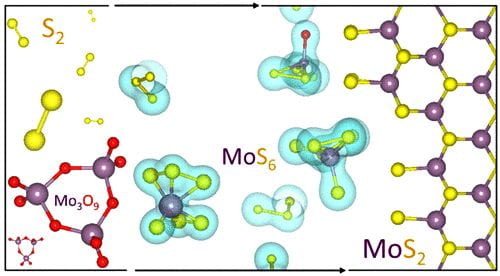 Scientific studies describing the most basic processes often have the greatest impact in the long run. A new work by Rice University engineers could be one such, and it’s a gas, gas, gas for nanomaterials.
Scientific studies describing the most basic processes often have the greatest impact in the long run. A new work by Rice University engineers could be one such, and it’s a gas, gas, gas for nanomaterials. Electronic transport through a
Electronic transport through a