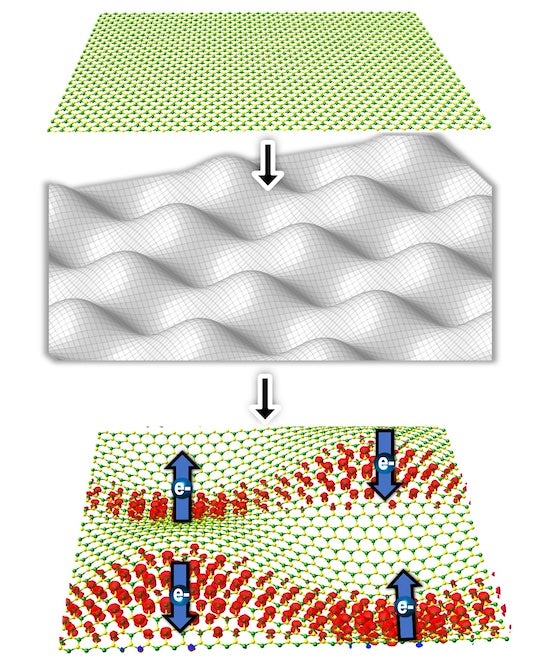
Representation of electron spins in a bent 2D material exhibiting the persistent spin helix structure. (Image courtesy of Sunny Gupta)
Wrinkles can be an asset — especially for next-generation electronics. Rice University scientists have discovered that tiny creases in two-dimensional materials can control electrons’ spin with record precision, opening the path to ultracompact, energy-efficient electronic devices.
If most devices today use the charge of electrons flowing through silicon to process and encode information, future computing may instead harness spin — a quantum property of electrons that takes on either an “up” or “down” value. Computing with spin could overcome the limitations of current silicon-based technology, reducing the energy footprint of devices and data centers at a moment when computing-driven energy use is soaring globally.
However, spintronics has to contend with a major challenge: Information encoded in spin is quick to decay and can be lost when the electrons in a material scatter and collide with atoms.
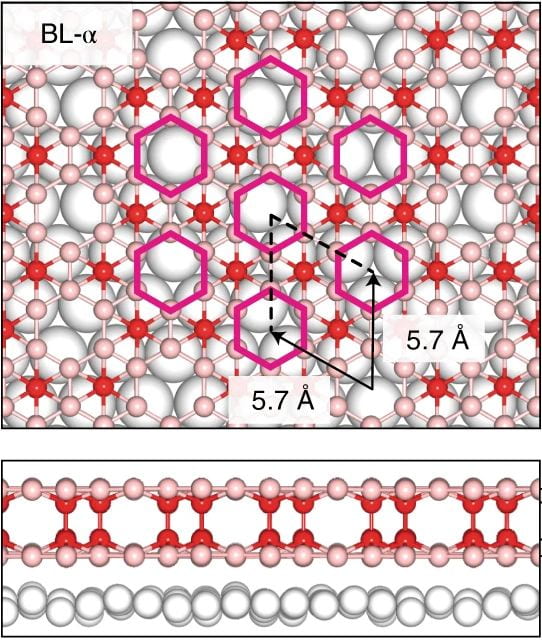
In a new study published in the journal Matter, Rice materials scientists have reported that bending atomically thin layers of materials like molybdenum ditelluride, MoTe2, gives rise to a unique spin texture called persistent spin helix, or PSH, which can preserve spin state even in scattering collisions.
– See more at Rice News





 contoured substrates
contoured substrates
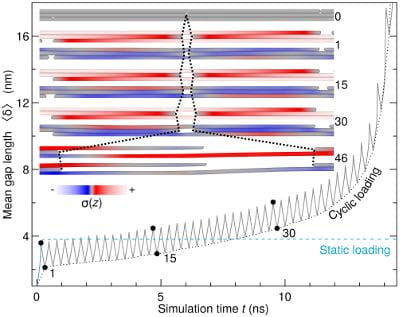 Up here in the macro world, we all feel fatigue now and then. It’s the same for bundles of
Up here in the macro world, we all feel fatigue now and then. It’s the same for bundles of 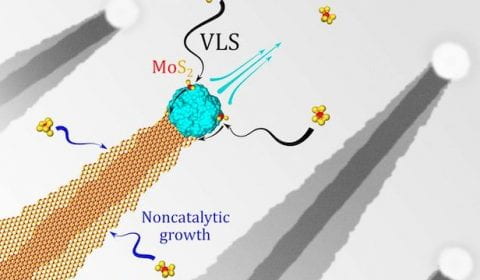 It’s now possible to quickly make ultrathin nanoribbons of
It’s now possible to quickly make ultrathin nanoribbons of 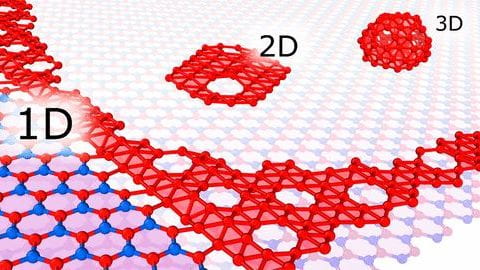
 If one layer of
If one layer of